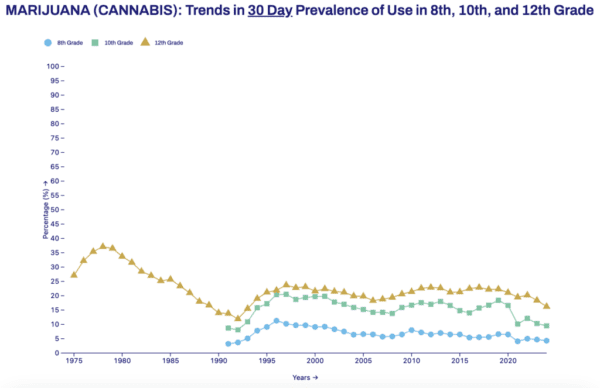
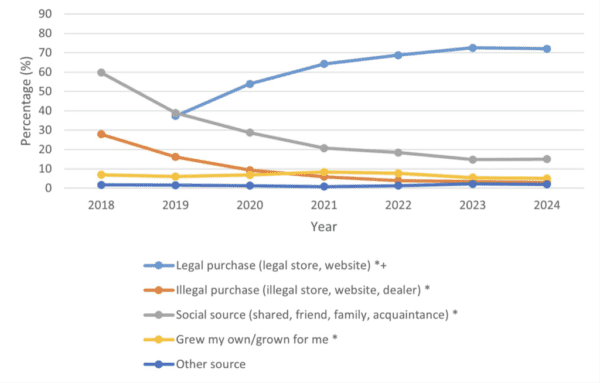
Science & Health Archives – Marijuana Moment Read More [[{“value”:” Even as more states have legalized marijuana, rates of teen use are on the decline. There was also a significant […]
Science & Health Archives – Marijuana Moment Read More [[{“value”:” As the marijuana legalization movement has expanded—and psychedelics policy has increasingly entered the mainstream conversation—a new survey shows that Americans […]
Science & Health Archives – Marijuana Moment Read More [[{“value”:” For the first time ever, researchers are administering LSD to patients in a Phase 3 clinical trial. The new study […]
Science & Health Archives – Marijuana Moment Read More [[{“value”:” New government-funded research on terpenes produced by the cannabis plant finds that some of the aromatic compounds could be promising […]
Science & Health Archives – Marijuana Moment Read More [[{“value”:” A new scientific review finds that high-CBD, low-THC cannabis products have potential to “significantly improve the quality of life” for […]
Science & Health Archives – Marijuana Moment Read More [[{“value”:” Cannabis-infused vaginal suppositories seem to reduce sexual pain in women after treatment for gynecological cancer, according to a new study. […]
Science & Health Archives – Marijuana Moment Read More [[{“value”:” Six years after Canada legalized marijuana sales nationwide, a new government report shows that daily or near-daily use rates by […]
Science & Health Archives – Marijuana Moment Read More [[{“value”:” Newly published research on the psychiatric effects of psilocybin suggests a complicated, potentially bidirectional relationship between the psychedelic’s therapeutic effects […]
Science & Health Archives – Marijuana Moment Read More [[{“value”:” A newly published report in the Journal of the American Medical Association (JAMA) finds that psilocybin-assisted therapy in a group […]
Science & Health Archives – Marijuana Moment Read More [[{“value”:” Giving dogs a dose of CBD could help counteract the ill effects of accidental THC consumption, according to a new […]
Science & Health Archives – Marijuana Moment Read More [[{“value”:” New research on how people cope with challenging psychedelic experiences suggests that some ways to manage a bad trip may […]
Science & Health Archives – Marijuana Moment Read More [[{“value”:” A new study on prescription opioid use in Utah following the state’s legalization of medical marijuana suggests the availability of legal […]