Health and Science
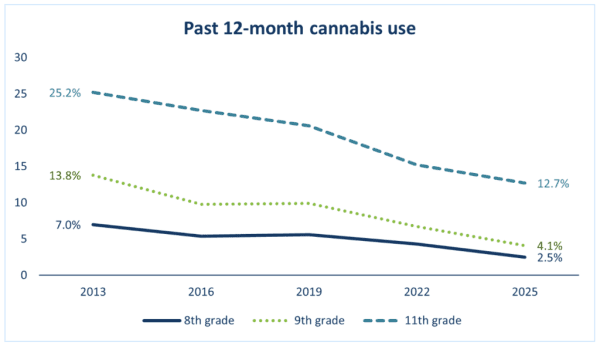
Despite concerns from marijuana legalization opponents who claimed the policy would lead to skyrocketing use by teens, cannabis consumption by middle and high school students in Minnesota is lower now than it has ever been over the past decade, according to newly published state data. “There continues to be a steady decline in youth cannabis
The post Teen Marijuana Use Is Lower Now Than Before Legalization, Government Study From Minnesota Officials Shows appeared first on Marijuana Moment.
There’s been a new breakthrough in the development of a marijuana breathalyzer, with a study partly funded by the Justice Department showing a potential pathway for a “portable, low cost” device that looks like an inhaler for asthma, built with 3-D printed material that can detect delta-9 THC without secondary lab analysis. The study, led
The post Federally Funded Study Reveals Marijuana Breathalyzer Breakthrough With 3-D Printed Roadside Tool Able To Detect THC appeared first on Marijuana Moment.
Alcohol and tobacco cause far more harms to people who consume them, and to society overall, than marijuana does, according to a new government-funded study. A panel of 20 experts with professional experience with substance use issues from across Canada were asked to evaluate 16 different drugs on 10 dimensions of harm to consumers, as
The post Alcohol And Tobacco Are More Damaging To Users And Society Than Marijuana Is, Government-Funded Study Concludes appeared first on Marijuana Moment.
Marijuana May Be A ‘Gateway To Women’s Orgasm’ In Sexual Health Treatment, Scientific Analysis Finds
Marijuana may be a “gateway,” but not in the stigmatized way it’s been portrayed by prohibitionists as a stepping stone to other drugs. Rather, a growing body of scientific literature signals cannabis is a “gateway to women’s orgasm” that could hold significant therapeutic potential in the treatment of female orgasmic disorder/difficulty (FOD), a new research
The post Marijuana May Be A ‘Gateway To Women’s Orgasm’ In Sexual Health Treatment, Scientific Analysis Finds appeared first on Marijuana Moment.
Getting the “munchies” after using marijuana isn’t just in your head. It’s a real biological phenomenon that could hold important, real-world implications for people suffering from conditions associated with appetite loss, according to a new study that also identified some of the most commonly desired food items while high. Researchers at Washington State University (WSU)
The post Scientists Reveal What Types Of Food The Marijuana ‘Munchies’ Make You Crave The Most appeared first on Marijuana Moment.
Artificial intelligence (AI) chatbots are surprisingly good at mimicking human psychedelic experiences, according to a new study in which researchers virtually dosed large language models (LLMs) such as ChatGPT with simulations of drugs like psilocybin, DMT and ayahuasca. For the study, researchers at the University of Haifa and Bar-Ilan University ran analyses that compared self-reported
The post AI Models Like ChatGPT Can Generate ‘Convincingly Realistic’ Psychedelic Experiences When Virtually Dosed, Study Shows appeared first on Marijuana Moment.
A new study suggests that cannabis-infused beverages may help reduce the harms of alcohol drinking by providing an alternative for consumers. Public health researchers have long searched for realistic ways to reduce the toll of alcohol, a substance linked to nearly 200 diseases and injuries worldwide, and the new paper sheds light on the role
The post Cannabis-Infused Drinks Offer Consumers A ‘Harm Reduction’ Alternative To Alcohol, Study Shows appeared first on Marijuana Moment.
Nearly 10 million American adults microdosed psychedelic substances such as psilocybin, LSD or MDMA in 2025, according to a new analysis. The results of the RAND Corporation survey indicate that microdosing—which involves ingesting a small amount of a drug to improve mood and well-being without experiencing hallucinations or a full-scale trip—is a popular practice in
The post 10 Million US Adults Microdosed Psychedelics Last Year, New Report Shows appeared first on Marijuana Moment.
More than a third of popular U.S. hip hop and rap music videos referenced marijuana in 2024, according to a new government-supported study. Artists like Snoop Dogg and Dr. Dre have helped drive that trend by promoting a “chilled” lifestyle, the researchers said. According to the analysis—which was funded by the Ministry of Justice and
The post More Than A Third Of Rap And Hip Hop Music Videos Feature Marijuana, Government-Funded Study Shows appeared first on Marijuana Moment.
Cannabidiol (CBD), a non-intoxicating compound in marijuana, “holds substantial promise as an anti-tumor agent” in addition to its other anti-inflammatory properties, a new scientific review Scientists explored CBD’s effect on many types of cancer—including some of the most aggressive ones, such as glioblastoma, which affects the brain. They also noted it can help suppressing the
The post CBD Has ‘Substantial Promise’ To Combat Tumors From Cancer, Scientific Review Shows appeared first on Marijuana Moment.
Laws aimed at preventing marijuana-impaired driving in almost 20 states are causing innocent people who show no signs of impairment to be criminalized and imprisoned for allegedly operating vehicles while under the influence, a new government-funded study shows. Lawmakers and regulators aiming to reduce drug-impaired driving have long sought to apply a familiar strategy from
The post Marijuana Users Are Being Unjustly Jailed For Allegedly Driving Under The Influence, Government-Funded Study Shows appeared first on Marijuana Moment.
Marijuana has therapeutic benefits for adults with disabilities and is commonly utilized to manage pain and other symptoms, a new government-funded study shows. “Cannabis use is prevalent across disability domains and the majority of respondents note they use cannabis to help with pain and to relax or relieve tension,” the researchers wrote. More than one
The post Medical Marijuana Use Is ‘Prevalent’ Among People With Disabilities—And It Helps Treat Pain, New Federally Funded Study Shows appeared first on Marijuana Moment.