Health and Science
Science & Health Archives – Marijuana Moment Read More [[{“value”:” A new review of scientific research on the lesser-known cannabinoid cannabigerol (CBG) says the compound has the “potential to modulate […]
Science & Health Archives – Marijuana Moment Read More [[{“value”:” A new study of more than 5,000 men whom researchers evaluated over the course of 44 years found “no significant […]
Science & Health Archives – Marijuana Moment Read More [[{“value”:” After years of delays, researchers are set to move forward on a landmark clinical trial meant to evaluate the efficacy […]
Science & Health Archives – Marijuana Moment Read More [[{“value”:” A newly published review of research on psychedelics and tobacco use concludes that there’s “evidence that psychedelics, in particular psilocybin, […]
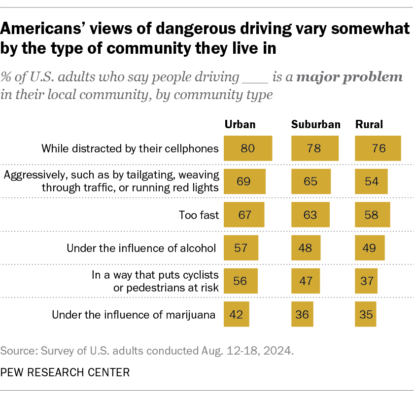
Science & Health Archives – Marijuana Moment Read More [[{“value”:” Americans view driving under the influence of marijuana as one of the issues they’re “least concerned” about when compared to […]
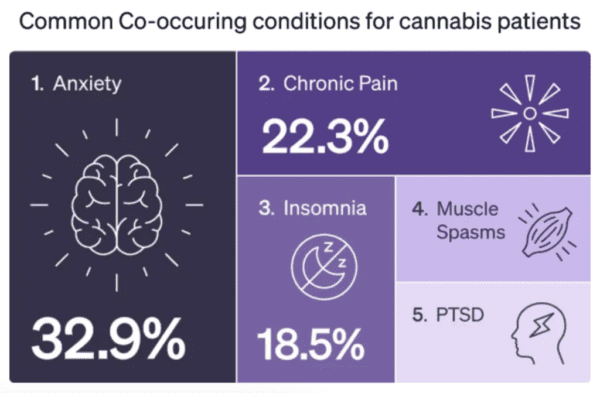
Science & Health Archives – Marijuana Moment Read More [[{“value”:” Recently published data from tens of thousands of medical marijuana patients across the U.S. show that pain, anxiety and post-traumatic […]
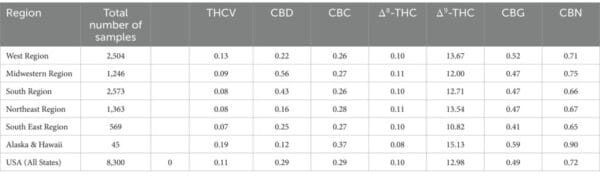
Science & Health Archives – Marijuana Moment Read More [[{“value”:” A new study led by the one of the only people permitted by the U.S. government to grow marijuana for […]
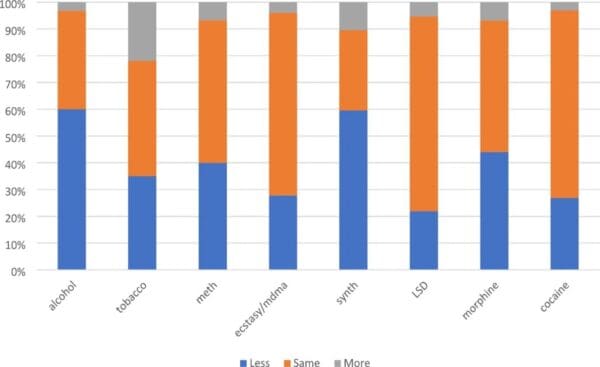
Science & Health Archives – Marijuana Moment Read More [[{“value”:” A new study on the impact of marijuana consumption on people’s use of other drugs suggests that for many, cannabis […]
Science & Health Archives – Marijuana Moment Read More [[{“value”:” A newly published study of product packaging from the commercial marijuana industry concludes that the market shift toward vape pens […]
Science & Health Archives – Marijuana Moment Read More [[{“value”:” Newly published research that combines an academic literature review with a survey of university students concludes that marijuana is likely […]
Science & Health Archives – Marijuana Moment Read More [[{“value”:” A pair of newly announced government grants totaling more than half a million dollars will fund research into hemp, marijuana […]
Science & Health Archives – Marijuana Moment Read More [[{“value”:” A new study finds that people who have used multiple different formulations of psilocybin—including whole mushrooms, mycological extract and a […]